#Biotechnology #FDAApproval #ClinicalTrials

www.nasdaq.com
Weekly Buzz: GSK's Lynavoy Gets FDA Nod; Reproxalap Rejected Again; LNSR Ends Merger Deal
(RTTNews) - This week's biotech landscape witnessed key FDA approvals, rejections, NDA resubmissions, merger terminations, trial discontinuations, and clinical trial data readouts across therapeutic areas such as Turner Syndrome, Late-Line Colorectal Cancer, Obesity, and Type 1 Diabetes.
#AnimalTesting #FDA #ResearchAlternatives

www.yahoo.com
The push to end animal testing is gaining steam, but technology can't fill the gap yet
Add Yahoo as a preferred source to see more of our stories on Google.
A social media post from the US Food and Drug Administration this week shows a big-eyed macaque staring out from behind bars.
#RareDiseases #MPS #FDAApproval

www.newsweek.com
Moms stage "funeral" outside FDA in drug protest
More than 100 mothers and advocates affected by a type of rare disease gathered outside the Food and Drug Administration (FDA) on Wednesday to stage a funeral to "mourn the futures" of their children, following the agency's rejections of various rare disease treatments.
#RareDiseases #FDAApproval #PatientAdvocacy
www.newsweek.com
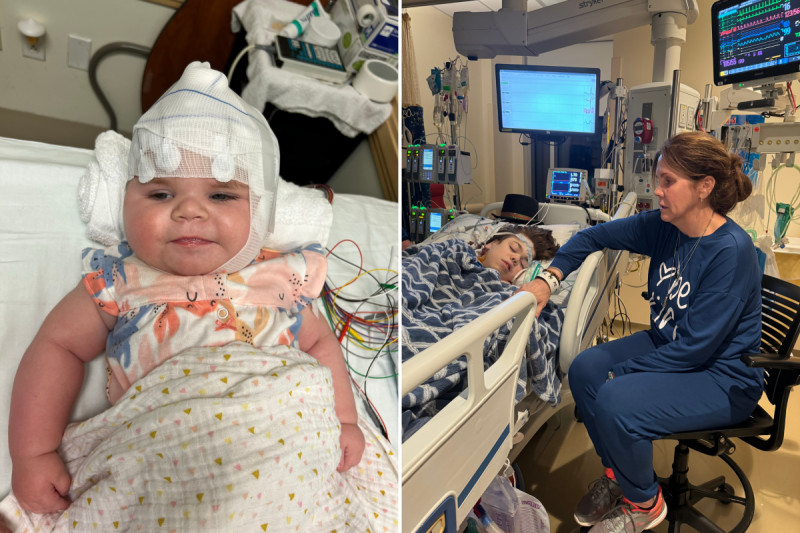
Mothers "terrified" as children could lose treatments after FDA denials
Mothers from across the country have spoken to Newsweek about their concern for their children after the Food and Drug Administration (FDA) has been rejecting various treatments for rare diseases that can be life-threatening.
#FDA #RareDiseases #HealthPolicy

www.newsweek.com
Republican launches investigation into Trump admin FDA denials
Senator Ron Johnson has launched an investigation into the Food and Drug Administration (FDA) over the agency's recent denials of treatments for rare diseases, the Wisconsin Republican said in an interview with Bloomberg.
#FDA #Vaccines #DrugRegulation

www.cbsnews.com
Trump administration's embattled FDA vaccine chief is leaving again
The Food and Drug Administration's embattled vaccine chief, Dr. Vinay Prasad, is once again leaving the agency -- the second time in less than a year that he's departed after controversial decisions involving the review of vaccinations and specialty drugs for rare diseases.